LPC Setting Strategy for ~1% Failure Rate
2025-10-01
Introduction
“For the low-positive QC sample, we recommend that a concentration be selected that, upon statistical analysis, would lead to the rejection of an assay run 1% of the time.
In this way, the sponsor ensures that the assay is performing as expected and that subject samples are correctly evaluated.”
— U.S. FDA, Immunogenicity Testing of Therapeutic Protein Products (2019)
This recommendation defines the conceptual anchor for setting the Low Positive Control (LPC) in ADA assays. Statistically, it corresponds to designing the LPC such that approximately 1% of its measurements fall below the cut point (CP) — i.e., the “1% fail zone.”
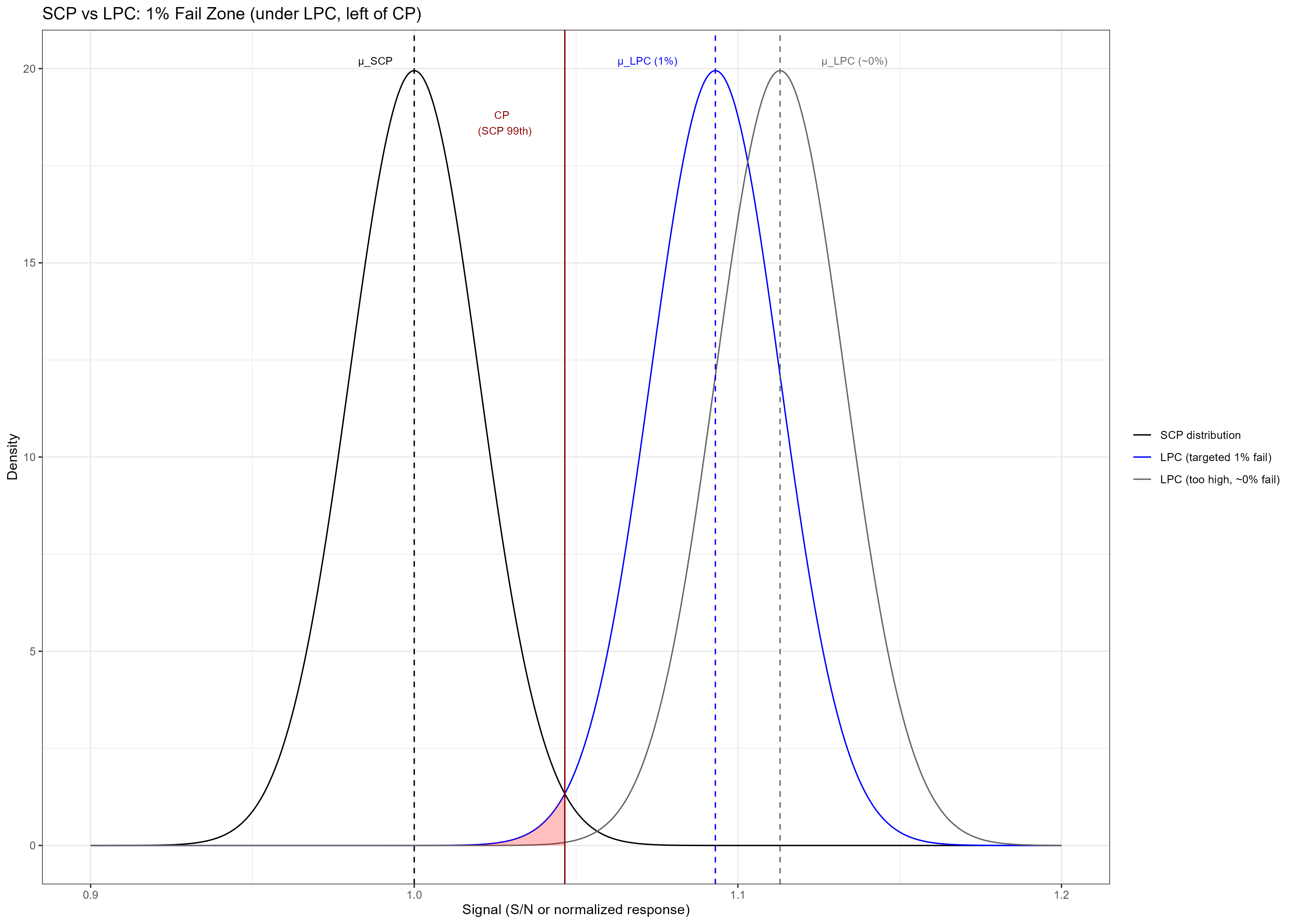
In practice, many laboratories now express the Screening Cut Point (SCP) in concentration units (rather than signal units) using calibration-based regression,and derive the LPC concentration by applying a one-sided t0.99 multiplier to the SCP estimate.
In the next section, we will examine:
- What this transformation means statistically,
- Why the current practice can underestimate or overestimate the real 1% failure probability, and
- How to interpret such deviation in Titer assay evaluations.